Conditions involving inflammation and fibrosis, including idiopathic pulmonary fibrosis
Celea Therapeutics (Celea) is a clinical-stage biopharmaceutical company dedicated to delivering transformative treatments for people with serious respiratory diseases. Its lead program, deupirfenidone (LYT-100), is a Phase 3-ready therapeutic candidate with the potential to establish a new standard of care (SOC) for the treatment of idiopathic pulmonary fibrosis (IPF) and other fibrotic lung diseases.
Deupirfenidone is an investigational drug not approved by any regulatory authority.
1 Raghu, Ganesh, et al. “Incidence and prevalence of idiopathic pulmonary fibrosis in US adults 18–64 years old.” European Respiratory Journal 48.1 (2016): 179-186
2 Raghu, Ganesh, et al. “Idiopathic pulmonary fibrosis in US Medicare beneficiaries aged 65 years and older: incidence, prevalence, and survival, 2001–11.” The lancet Respiratory medicine 2.7 (2014): 566-572
3 Kreuter, Michael, et al. “Epidemiology, healthcare utilization, and related costs among patients with IPF: results from a German claims database analysis.” Respiratory Research 23.1 (2022): 62
4 Snell, N., et al. “P272 Epidemiology of idiopathic pulmonary fibrosis in the UK: findings from the British lung foundation’s ‘respiratory health of the nation’ project.” (2016): A236-A236
5 French guidelines PNDS
6 Rodríguez-Nieto, Maria Jesus, et al. “Economic burden of idiopathic pulmonary fibrosis in Spain: a prospective real-world data study (OASIS study).” PharmacoEconomics 41.8 (2023): 999-1010.
7 Iommi, Marica, et al. “Occurrence of idiopathic pulmonary fibrosis in Italy: latest evidence from Real-World data.” International Journal of Environmental Research and Public Health 19.5 (2022): 2510.
8 Fisher, M., Nathan, S. D., Hill, C., Marshall, J., Dejonckheere, F., Thuresson, P., & Maher, T. M. (2017). Predicting life expectancy for pirfenidone in idiopathic pulmonary fibrosis. Journal of Managed Care & Specialty Pharmacy, 23(3-b Suppl), S17–S24. https://doi.org/10.18553/jmcp.2017.23.3-b.s17
9 Dempsey TM, Payne S, Sangaralingham L, Yao X, Shah ND, Limper AH. Adoption of the Antifibrotic Medications Pirfenidone and Nintedanib for Patients with Idiopathic Pulmonary Fibrosis. Ann Am Thorac Soc. 2021 Jul;18(7):1121-1128
10 FVC decline at 6 months was estimated assuming linear decline over time.
11 Valenzuela, C., Bonella, F., Moor, C., Weimann, G., Miede, C., Stowasser, S., & Maher, T. (2024, September). Decline in forced vital capacity (FVC) in subjects with idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF) compared with healthy references [Poster presentation]. European Respiratory Society International Congress, Vienna, Austria; and Luoto, J., Pihlsgård, M., Wollmer, P., & Elmståhl, S. (2019). Relative and absolute lung function change in a general population aged 60–102 years. European Respiratory Journal, 53(3), 1701812. https://doi.org/10.1183/13993003.01812-2017
12 Part B analysis is based on switch patients (those who completed 26 weeks of placebo or pirfenidone in Part A and then were re-randomized to receive deupirfenidone 825 mg TID in Part B). Patients were re-baselined to the last available FVC measurement obtained prior to the first administration of deupirfenidone 825 mg TID in Part B. Observed mean (SE) values are presented over time as of May 9, 2025.


 Patient Need
Patient Need  Milestones Achieved & Development Status
Milestones Achieved & Development Status 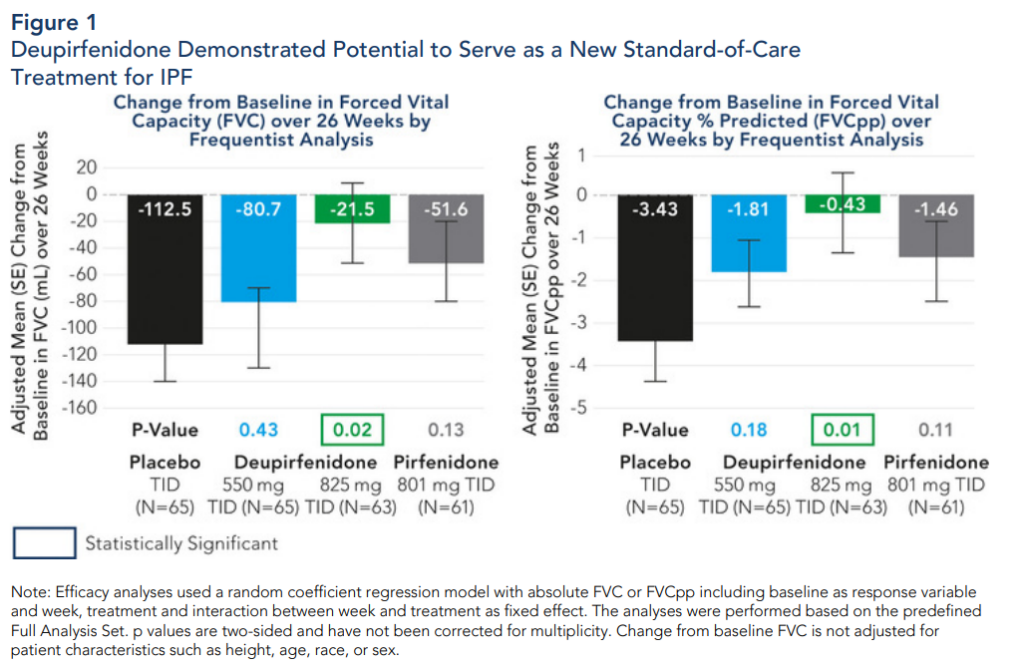
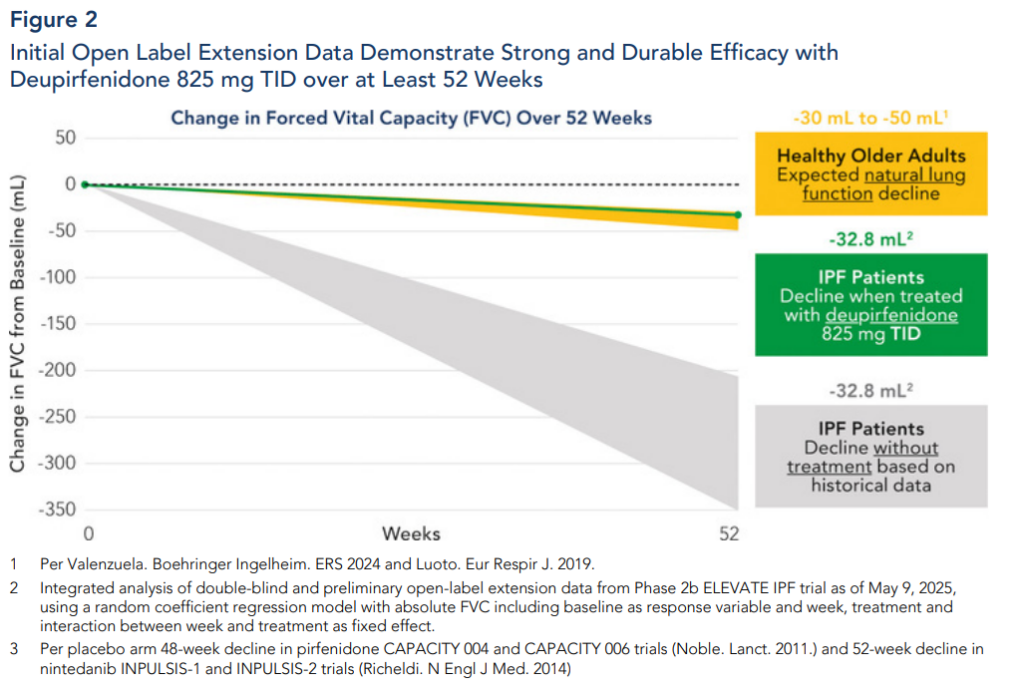
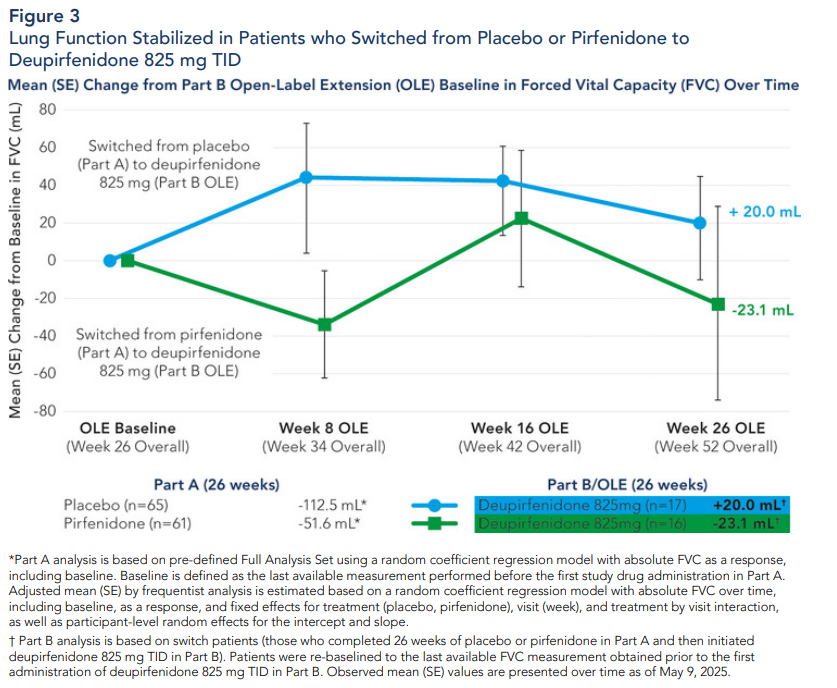
 Expected Milestones
Expected Milestones  Intellectual Property
Intellectual Property 
 Press Releases
Press Releases  Latest Publications
Latest Publications